|
Water contains Nitrogen, Oxygen, Carbon dioxide and other gases. Total Pressure of these dissolved gases is equal to the sum of partial pressures of all gases dissolved in
water.
PTDG = pO2 + pN2 + pCO2 + pH2O + p…
Where:
PTDG = Total Dissolved Gas Pressure
pO2 = partial pressure of Oxygen
pN2 = partial pressure of Nitrogen
pCO2 = partial pressure of Carbon dioxide
pH2O
= partial pressure of water vapor
p … = partial pressure of all other dissolved gases like hydrogen sulfide and methane
The total amount of gas dissolved in water = The difference between sum of partial pressure of all gases in air or Barometric pressure and sum of partial pressure
of all gases in water. This can also be written as Percent saturation of the gases in water.
or PTDG(%) = (PTDG / PBP) x 100
where PTDG(%) = Total Gas Pressure expressed as percent saturation
PBP = Barometric Pressure at water surface
When the partial pressure of a gas in water equals to its partial pressure in the air, there is no net movement from air to water or vice versa. Then, the gases in
air are said to be in equilibrium with the dissolved gases in the water is in equilibrium with air:
If the partial pressure of a gas in water is less than its partial pressure in the air, then the water is under-saturated. So, that gas will move from air to
water.
If the partial pressure of a gas is less in the air than its partial pressure in water, then water is supersaturated with that gas. The gas will diffuse from water to
atmosphere.
It is possible for water to be under-saturated with one gas but supersaturated with another..
In supersaturated water, the gases that are supersaturated are always diffusing into the atmosphere in the form of bubbles. If fish are exposed to this water, the gas
will diffuse across the gills into the fish. As blood gets saturated with gases, they start escaping in the form of bubbles. Bubbles can form in the gills, fins, skin and blood. If bubbles are formed in blood, it can block
circulation, especially to small fish. Some of the symptoms of gas supersaturation are pop-eye, floating upside down, bleeding around the eyes, swollen belly and secondary bacterial and fungal infections caused by stress. Sometimes
there may be no symptoms but when you run your hand in water, you may see a swarm of micro bubbles. Supersaturation may appear and disappear randomly depending on the pumping rate and aeration in the system.
If Total dissolved gases (PTDG) is 110% at the surface of a water, the PTDG saturation 3 feet below the surface is only 100%. For every 3 feet depth, because the water pressure goes up (hydrostatic pressure), the PTDG saturation actually experienced by the fish drops by 10%. Therefore, when the surface PTDG is 120%, in a pond that is 6ft deep, fish at the bottom are exposed to a PTDG level of only 100%.
Fish sense high gas pressures, and like a diver, will go deeper in the pond to compress the gases to prevent bubble formation.
Super saturation resistance is species, gas and environment specific. For instance, inert gases such as Argon, Radon and Nitrogen are particularly harmful, as most fish
are not equipped to cope with these gases. Nitrogen and argon typically become supersaturated as a result of rapid temperature increases (greater then 6*C) or entrapment of air under pressure (eg. aeration at depths greater then 6
ft of water, saturated well water pumped from depth, hole in suction side of pump).
Supersaturation is usually not a problem if the water source for the pond is surface water (stream, river, municipal water). However, if surface water has dense
growths of algae, plants (lakes and ponds), water can be supersaturated with dissolved oxygen on bright, sunny summer afternoons. If water intake is from right under the dam falls, water could be supersaturated with oxygen.
Groundwater almost always contains high amounts of dissolved Carbon dioxide and Nitrogen. Heavily planted ponds or algae blooms in source water or the pond itself can
produce dangerous amounts of carbon dioxide at night or on cloudy days. In recirculating systems, the carbon dioxide produced by the fish, plants and other living things in the pond must be removed. Or else, it will dissolve in
water to become Carbonic acid and lower the pH. In general, Carbon dioxide levels above 20 ppm can lead to stress and lead to death above levels of 30-40 ppm.
CO2 + H2O H2CO3
Fish cannot completely digest carbohydrates (wheat, cereal, oats, soybean, corn) and fibers present in the feed. When these undigested
sugars and starches pass through the gut, good bacteria living in the colon use them as food. This process produces gases like Nitrogen, Oxygen, Hydrogen, Carbon dioxide and Methane. Methane can occur in the sediment of deep lakes or ocean bottoms or deep wells where high nutrient levels and anoxic conditions are
prevalent.
Foods high in sulfur like broccoli, Brussels sprouts, cauliflower,
meat, eggs, cheese, milk, garlic, onions, horseradish can produce hydrogen sulfide during digestion. Hydrogen Sulfide can also occur in the sediment of deep lakes or ocean bottoms or deep wells where high nutrient and
sulfate levels occur along with anoxic conditions. In the absence of oxygen, certain bacteria can reduce sulfate ions, producing hydrogen sulfide as a by-product. You may have witnessed the presence of hydrogen sulfide in your pond
while repotting your waterlilies or spring-cleaning your bead filters or while backwashing the EZPod after a long time. Hydrogen sulfide is highly toxic to aquatic animals. Many fish cannot tolerate levels greater then 0.003 mg/L.
Hydrogen sulfide is highly soluble in water making it impractical to use air stripping as a means of removal. Alternatively, water change or addition of ozone can be used to reduce/destroy hydrogen sulfide.
Ammonia is a common by-product of fish metabolism. As a general rule, approximately 3% - 4.5% ammonia is produced per Kg of feed consumed. Un-ionized ammonia is highly
toxic to fish. Ammonia can also occur in groundwater supplies either as naturally occurring concentrations or as a result of pollution from septic systems, agriculture or, municipal discharges. Ammonia is highly soluble and like
Hydrogen Sulfide, cannot be air stripped. Addition of Ozone will oxidize nitrite but will not affect un-ionized ammonia. The only way to remove Ammonia is have an efficient bio-filtration.
Supersaturation can be a problem even in winter, when most plants are dormant, algae are dead and fish resting at their basal metabolic rate. Source water from stream,
river and reservoir may be supersaturated with Carbon dioxide if the surface is iced up and the resident fish are gathered near the water intake. Another scenario is addition of very cold water during water change. Cold water has
lot of dissolved gases. Municipality pumps force even more air into water while pumping with great pressure. If large water changes are done with cold water in a single stretch, it can result in supersaturation. This can be
prevented by doing the large water change with several breaks; mixing cold water with hot water and letting it fall from a hose from a height. If the pond is enclosed, but aerated, it can still be supersaturated with Carbon
dioxide, if the air pump is sucking air within the enclosure. Presence of plants can make it worse. Installation of an air exchanger or placing the air pump outside the pond enclosure can prevent this problem.
Water can be supersaturated when heated in a closed boiler. As temperature increases, solubility of gases decreases but in a closed boiler, gases cannot escape
completely. Water will be supersaturated. This usually happens in winter when ground water is heated before adding. If adding municipal water is heated in a boiler and added to an indoor pond, the problem is worse. Chlorine from
hot water will escape as gas rather than mixing with dechlorinators, filling the room with Carbon dioxide which in turn will be sucked by air pumps.
Increasing water depth increases the gas carrying capacity of water. The greater the depth at which air is injected, the greater the super saturation will be.
Air-diffusers placed at great depths can cause supersaturation of water. Similarly, pumping water from deep wells can often result in water saturated with one or more gases. Once the water is pumped to the surface, the reduction in
pressure can cause these gases to become supersaturated.
Faulty plumbing can also lead to supersaturation. Air is sucked into the water through small, “pin-hole” leaks in the plumbing fittings and pipes on the
suction (inlet) side of pumps. As the entrained air passes through the pump, it is pressurized and some of the gases are driven into water. More gas entrainment can occur on the pressure (outlet) side of a pump, particularly across
a partially-open, true-union, PVC ball valve. Cavitating pumps that create turbulent air in the water are a common cause of supersaturation.
Even well water can be supersaturated if air is sucked into water when the well surges. Surging occurs when sands dislodged during well drilling partially clog the
screen or strainer at the bottom of the well casing. This impedes the flow of water into the casing, which results in water being pumped out of the well faster than it flows into the casing from the aquifer. When the water level
inside the casing drops below the bottom of the intake pipe strainer, air is sucked into the line and gases are forced into solution as water passes through the pump. This problem is most common in new wells. Screens of older wells
in areas with hard, alkaline groundwaters may become clogged by encrustations of lime or iron oxide. Cleaning with muriatic (dilute hydrochloric acid will correct this problem and most well-drillers do this routinely.
Note: Air bubbles entrained in the water by leak in the plumbing or well surging makes a hissing sound as the water travels along the piping. This sound is often
most noticeable at elbows or “T” fittings.
Preventing Supersaturation
- Proper plumbing and well maintenance can eliminate many of the situations faced with well water.
- Aerating the ground water/well water in a holding tank before adding is good practice. Adding water with a garden hose/ mist sprayers or in the form of a waterfall can
degas water.
-Cleaning pipe fittings carefully with an approved solvent before gluing will remove biodegradable particles present on the fittings (which otherwise will degrade over
time leaving pinholes.
- Apply generous amount of adhesives to both pipe and fitting and join them with a quarter twist of pipe against fitting. Apply teflon tape or pipe compound liberally to
the threads of reducer bushings or other threaded fittings, particularly at the pump inlet and outlet.
- Do not plant excessively. Add colorants or put a shade cloth over the pond to reduce algae.
- Run air pumps 24 x 7
- Incorporate streams, waterfalls, arc falls or mist makers in the pond.
- Make small water changes or a large water change with many breaks.
- Avoid static media that needs regular cleaning (EZPod, Trickle Tower, bead filter, sand filter), if you do not clean them regularly.
- Use Pond potting soil to pot water plants. Avoid gravel bottom/gravel streams.
Degassing
Air pumps, though very cheap, are very efficient in aerating/degassing. The introduction of air bubbles removes Carbon dioxide at about the same rate as it adds
Oxygen. Because of this, aerators maintain Carbon dioxide levels below 10 mg/l. Pure oxygen and pressurized oxygen/ozone delivery systems on the other hand, are very effective at aeration but do not remove Carbon
dioxide. So Carbon dioxide accumulates in such systems and lowers the pH. In such cases, degassing systems like packed columns or spray towers using a high rate of fresh air counter-flow are used to degas Carbon dioxide
from the system.
Packed Columns tall, round columns with >6” diameter, packed with Bio-barrels or other perforated media that allow water to pass through freely. They degas
water if the water is supersaturated or aerate if the incoming water has low oxygen levels. The following graph helps determine the length of the column, if you know the final concentration of dissolved oxygen you need at your
water temperature.
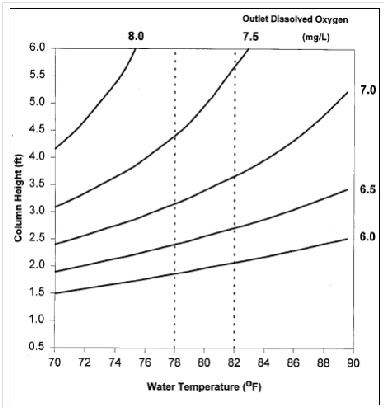
For example, if the water temperature ranges from 78*F to 80*F and you want to saturate the water with 90% dissolved
oxygen (between 7.0 to 7.3mg/L), then your column should be packed between 3.5ft to 4ft. You need a longer column at
higher temperatures. Remember to add about 2 feet to include plumbing inlet and outlets. The diameter of the column
depends the flow rate that your pump is pushing through. You can check the following table to help you select the size of air pump, diameter of the column and the size of media.
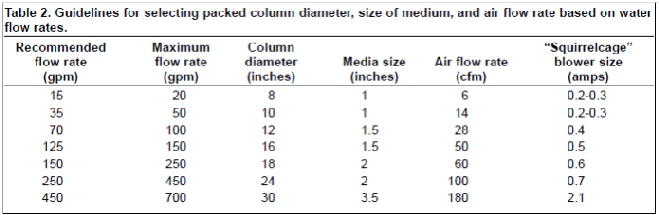
If higher flow rates are used, they can flood the column, preventing degassing. Columns smaller than 6” diameter can
have water flowing on the column walls rather than through the center. To degas Carbon dioxide, Hydrogen sulfide,
Methane, the column must be aerated with forced air (with an air pump). Air must be blown in the opposite direction of
water flow in order to break up the water molecules. This means the lower end of the column must be sealed or submerged in water. Otherwise, air will flow out of the column with water in the same direction.
Bibliography
John A. hargraves and Craign S. Tucker, Design and Construction of Degassing Units for Catfish Hatcheries, Southern Regional Aquaculture Center Pub. No
. 191
Colt, J. and G. Bouck. 1984. Design of packed columns for degassing.Aquacultural Engineering 3:251-273.
Hackney, G.E. and J.E. Colt. 1982. The performance and design of packed column aeration systems for aquaculture. Aquacultural Engineering 1:275-295.
Weitkamp, D.E. and M. Katz. 1980. A review of dissolved gas supersaturation literature. Transactions of the American Fisheries Society 109:659- 702.
|

